Flux Filtration for HCl Pickle Lines
You are here

Beta’s new Flux Filtration System extracts contaminants from flux tanks on HCl Pickle Lines ONLY. Our system works continuously, maintaining a solids-free flux tank.
- Decrease gross zinc consumption
- Minimize dross and ash production
- Improve product cosmetics
- Eliminate flux treatment or disposal
Overview:
The system removes iron contaminants carried into the flux tank from the rinse tanks at a processing rate of 6,500 – 10,000 gallons per day. The processing rate is dependent upon the solids load in the flux tank. If there are high levels of contamination in the incoming flux, solutions processed by this system should return the filtered flux solution with approximately half of the incoming concentration of ferrous. The targeted maximum concentration of iron ions in solution should reach ≤ 0.5 % by weight.
Components include: 1 or 2 large tanks, mixer, microfilters, filter press, air diaphragm pumps, instrumentation, framework, and control panel.
- Removing ferrous ions maintains a clean, efficient flux tank.
- Clean flux tanks save money. Saving just 1% in excess GZU results in a savings of $12,500 per million pounds of galvanizing.
- LME Zinc $1.20/lb. + $0.05 delivery = $1.25/lb
- Power consumption is similar to operating a household clothes dryer, < 3kWh per ton.
- The system requires minimal operator attention— approximately 30 minutes per shift.
Operation Diagram
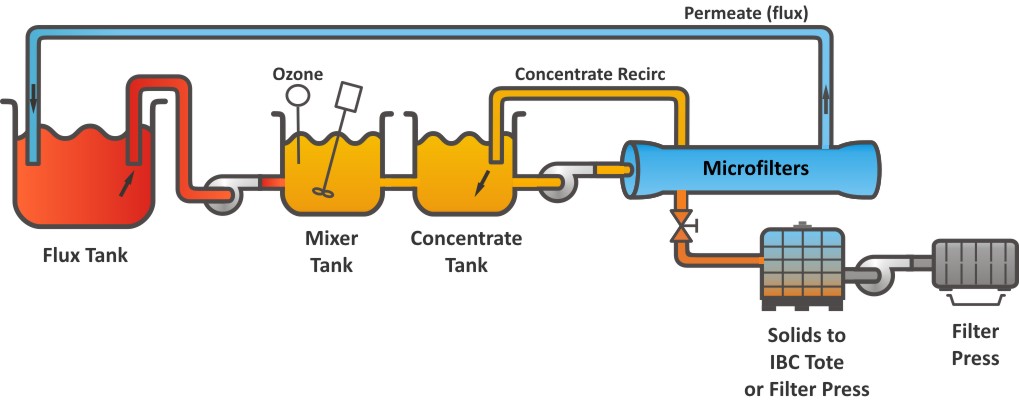
Operation steps
Step 1. The Flux Filtration System is impervious to pH and can run at elevated temperatures. However, the Flux Tank chemistry should be monitored regularly and balanced each week.
Step 2. Soluble Ferrous (Fe+2) ions are converted to insoluble Ferric (Fe+3) solids. Beta’s patent pending process continually produces highly oxidative Ozone and injects it into the flux filtration loop through our unique generation and injection system. There is no need to purchase or handle highly corrosive chemicals. In processes that contain sulfate contaminants, a concentrated solution of Barium Chloride is blended with the ozone and flux solution to convert and remove the sulfates as an insoluble suspended Barium Sulfate solid.
Step 3. Next, the solution enters the MicroFilter, a self-cleaning, crossflow filter.
Step 4. After Filtration, the solids are pumped through a Filter Press. The clean, contaminant-reduced filtrate is returned to the Flux Tank.
Step 4. Our flux filtration process runs continuously with very little attention. The process is self-cleaning and only requires minimal weekly cleaning.
Control Software
A computer or a control-panel-mounted HMI directs the operation of the system. Beta’s proprietary software allows the user to view live operating conditions and to initiate tasks manually or automatically.
The software contains set points which optimize the efficiency of the system.
Remote Monitoring - allows the operator, management, and Beta engineers to monitor the system from a computer or smart phone by means of a free app.
Wire & Cable magazine
Beta's Oxyfilter is featured in the Nov/Dec 2017 issue of Wire & Cable Technology International magazine. Check out our article which describes how the Oxyfilter recovers a galvanizer's flux tank.

